Getting the best from water-soluble fertilisers
Water-soluble fertilisers have become a key component of many nutritional regimes, due to their simplicity of application, cost-efficiency and rapid agronomic response. However, whilst they can easily be overcome, there are issues sometimes with preparation that can create problems for the uninitiated.
 Wendy Windscheffel, Technical Support Manager for Headland Amenity, says it’s worth noting some of the features that are common to all water-soluble products when using them for the first time.
Wendy Windscheffel, Technical Support Manager for Headland Amenity, says it’s worth noting some of the features that are common to all water-soluble products when using them for the first time.
Only so much product will actually dissolve.
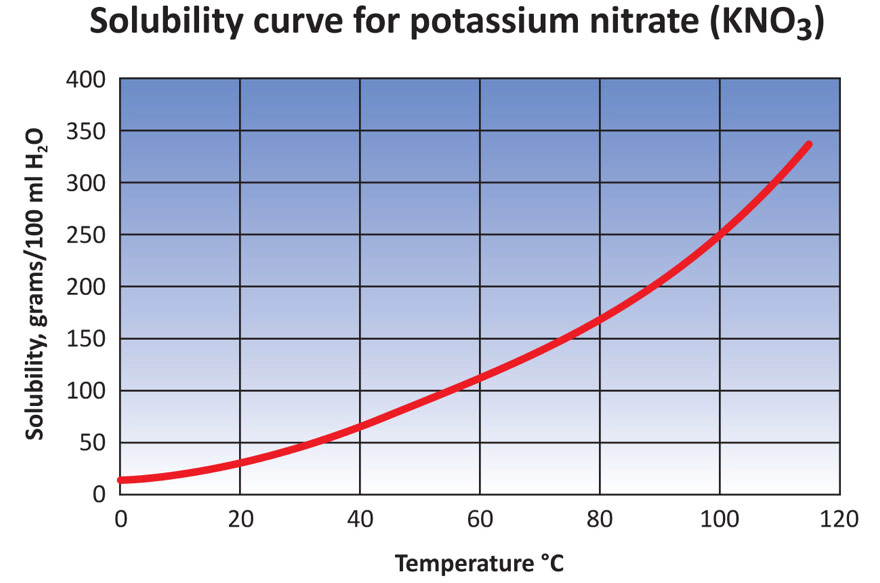
Each water-soluble fertiliser has a maximum solubility potential (a maximum amount of product that will solubilise in a given amount of water, before no more will dissolve). When this maximum amount has been reached, the solution is said to be ‘saturated’ and further addition only results in un-dissolved crystals, destined to block nozzles and filters.
This is one of the most common issues encountered whilst preparing a water soluble fertiliser, particularly if a small vessel like a bucket is utilised to dissolve the product. For example, we know that at 10ºC water temperature, the maximum amount of potassium nitrate that can be physically dissolved is 200g per litre. If we are using a 10L bucket of water, this means that we can dissolve 2,000g (2kg). At this point, the solution is ‘saturated’. If you were to add 5kg into the 10L bucket, 2kg would dissolve and 3kg will remain un-dissolved.







